

Cardiac
Research and development
Do you want to know the effects of your molecule, extract or compound on electric or contractile cardiac activity?
Do you want to qualify electrical or contractile cardiac activity of your transgenic animal model ?
We offer completing in vitro, ex vivo and in vivo assays to give you answers.
Safety pharmacology
Do you want a preclinical study on the effects of your molecule on cardiac repolarization ?
We offer GLP (Good Laboratory Practice) compliant assay recommended by the International Conference on Harmonisation.
Technology:
|
 |
Test 1 : In vivo exploration
- Cardiac electrical function: ECG
| Standard peripheral six-lead ECG is recorded on anesthetized animals under baseline conditions and after injection of pharmacological compounds.One-lead ECG is recorded by telemetry on conscious animals for continuous recordings on 24 or 48 h, day-night comparisons or the effects evaluation of pharmacological compounds.Measured parameters: RR interval, P wave, PQ interval, QRS complex and QT interval durations. ECG abnormalities (conduction blocks, premature beats, tachycardia…) are documented. |
- Intracardiac recording
This assay combines non-invasive (surface electrocardiogram) and invasive (catheter-mediated intracardiac) recording of spontaneous cardiac electrical activity as well as of cardiac responses to programmed electrical stimulation (PES).
It is designed to explore cardiac electrical activity and to evaluate arrhythmias
This technique can also be used to produce animal models of complete atrioventricular block by radiofrequency-mediated His bundle ablation.
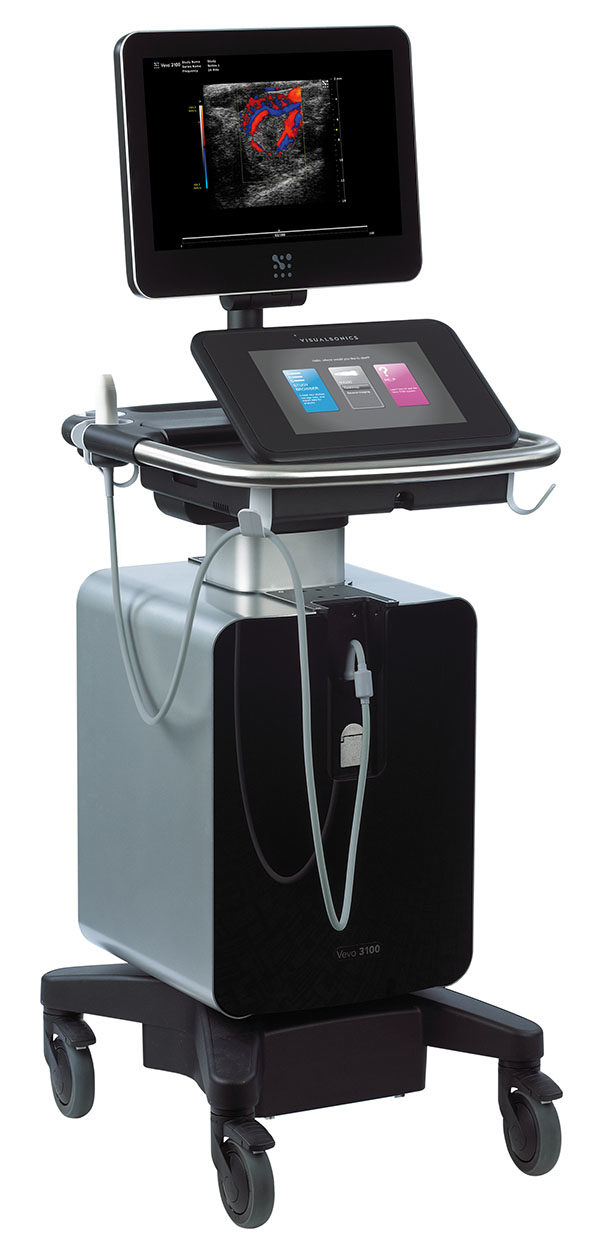
- Echocardiography Vevo 3100 (Fujifilm)
|
Biogenouest network, IBiSA network and FEDER funding Anesthetized mice or rats Assessment of systolic function, diastolic function and anatomical parameters… Cardiac dysfunctions and valvulopathies
|
 |
Cardiac pump function is evaluated in vivo by measuring left ventricular pressure and volume and constructing left ventricular pressure-volume loops. Measured parameters: heart rate, end diastolic and systolic left ventricular pressures and volumes. Peak left ventricular pressure (DP), maximum rate of increase in pressure during contraction (dP/dt max), maximum rate of decrease in pressure during relaxation (dP/dt min) are also available. |
Test 2 : Ex vivo exploration
- Cardiac contractile function: Isolated Working Heart
| This assay uses the isolated working heart technique to examine cardiac inotropy, chronotropy and pump function without the complications of an intact animal model but with an intact coronary perfusion system.Measured parameters: heart rate, peak left ventricular pressure (DP), maximum rate of increase in pressure during contraction (dP/dt max), maximum rate of decrease in pressure during relaxation (dP/dt min), cardiac output, cardiac heart cycle period and integrity stress…Those parameters are continuously recorded under vehicle and after increasing concentrations of a test item. |
- Cardiac contractile function: papillary muscles
This assay is designed to assess the direct effects of pharmacological compounds on contractility of isolated cardiac multicellular preparations.
Excised papillary muscles are placed in an experimental chamber, perfused with oxygenated physiological solution and paced at a constant rate. Contractions are recorded with a mechano-electrical force transducer.
Measured parameters: twitch amplitude (developed contractile force), twitch duration, time of half-maximum contraction (contraction velocity) and time of half-maximum relaxation (relaxation velocity).
Test 3 : In vitro exploration
- Action potential recording by intracellular microelectrode technique on perfused isolated tissues: auricular and ventricular myocard (mice, rat)
- Action potential and ionic current recording on isolated cardiomyocytes (mice, rat) by patch-clamp
- Functional characterization of ionic channels by patch-clamp on stable or transiently transfected cell lines [Nav1.5, Cav1.2, Kv7.1 (KvLQT1), Kv11.1 (ERG), Kir2.1, Kir6.2, HCN2...]
|
|
 |
Animal models:
- Septic shock
Animal model: rat
Method: intravenous injection of lipopolysaccharide of Escherichia coli. This model can be studied between 3 pm – 24 hours post-injection.
- Ischemic heart failure
Animal model: rat
Method: left coronary artery ligature with a study during the acute phase of myocardial infarction or later, up to 12 weeks post-ligature, with 2 levels of heart failure, compensated and decompensated.
- Progressive cardiac conduction disease
Animal model: heterozygous knock-out mice for the gene Scn5a which code the sodium channel Nav1.5.
- Dilated cardiomyopathy
Animal model: rat
induced by chronic treatment with Adriamycin



